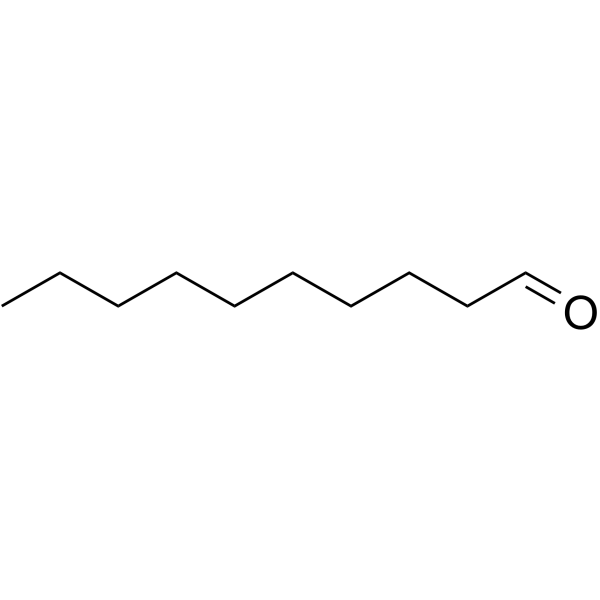
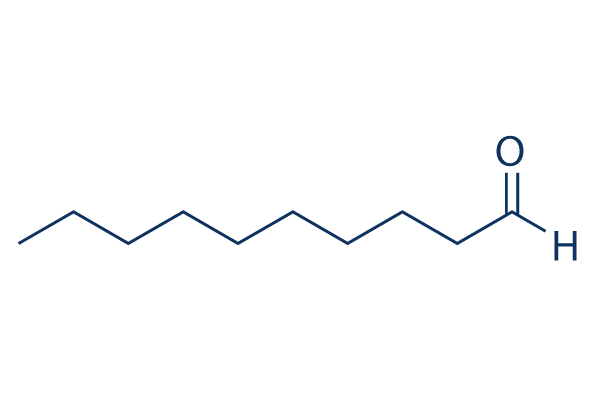
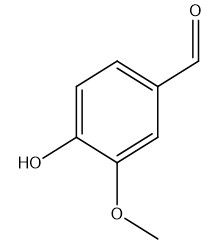
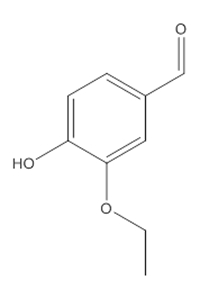
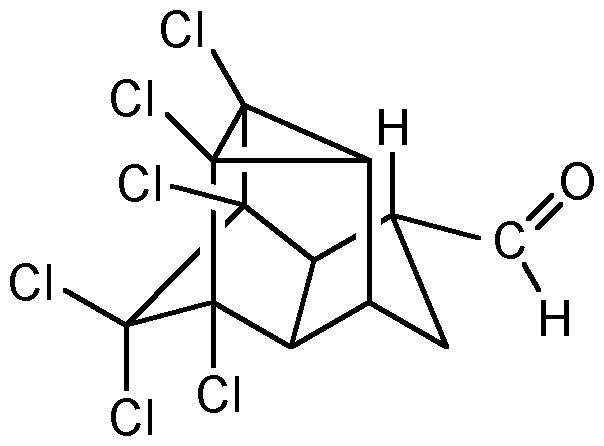
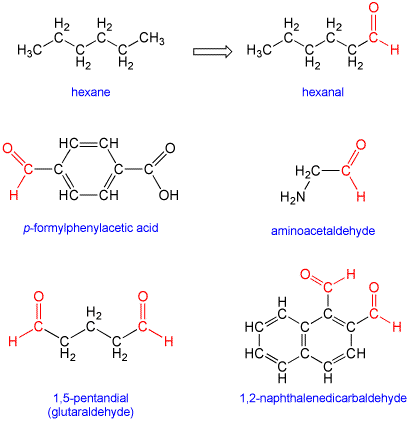
Continuous Flow Oxidation of Alcohols and Aldehydes Utilizing Bleach and Catalytic Tetrabutylammonium Bromide | Organic Process Research & Development

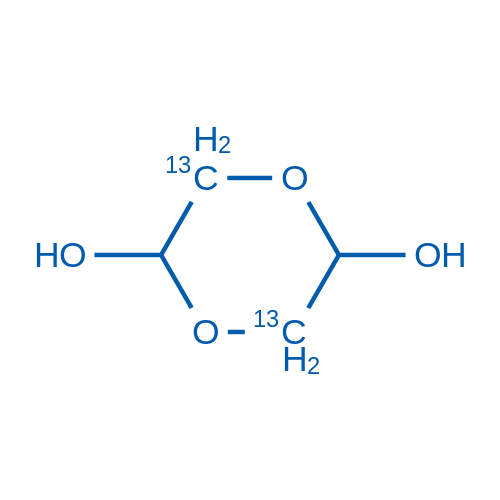
Aldehyde-catalysed carboxylate exchange in α-amino acids with isotopically labelled CO2 | Nature Chemistry
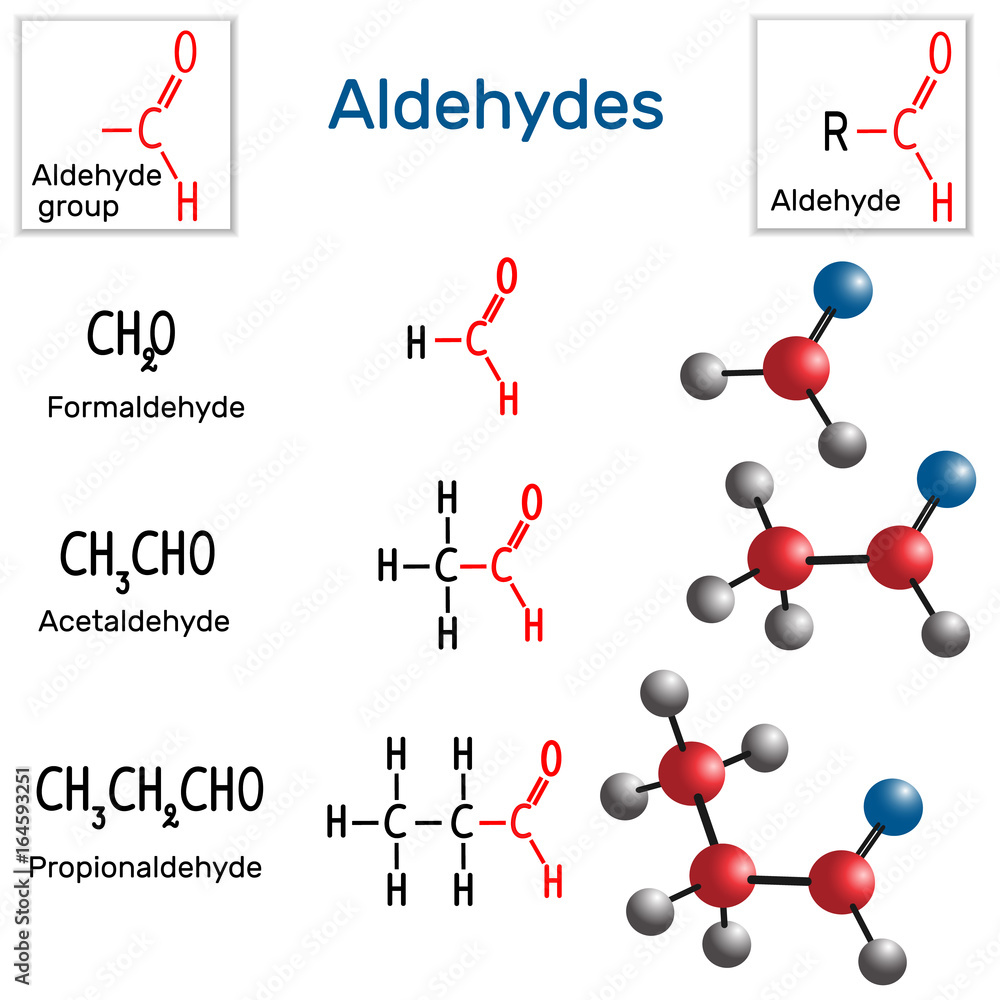
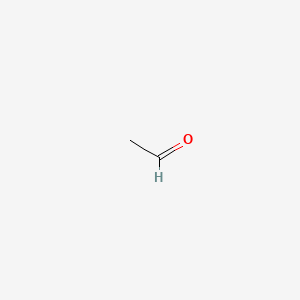
Aldehydes. Chemical formula and molecule model formaldehyde, acetaldehyde and propionaldehyde Stock Vector | Adobe Stock

An overview of synthetic N-acyl-ωaminoaldehydes. Molecular formulas of... | Download Scientific Diagram
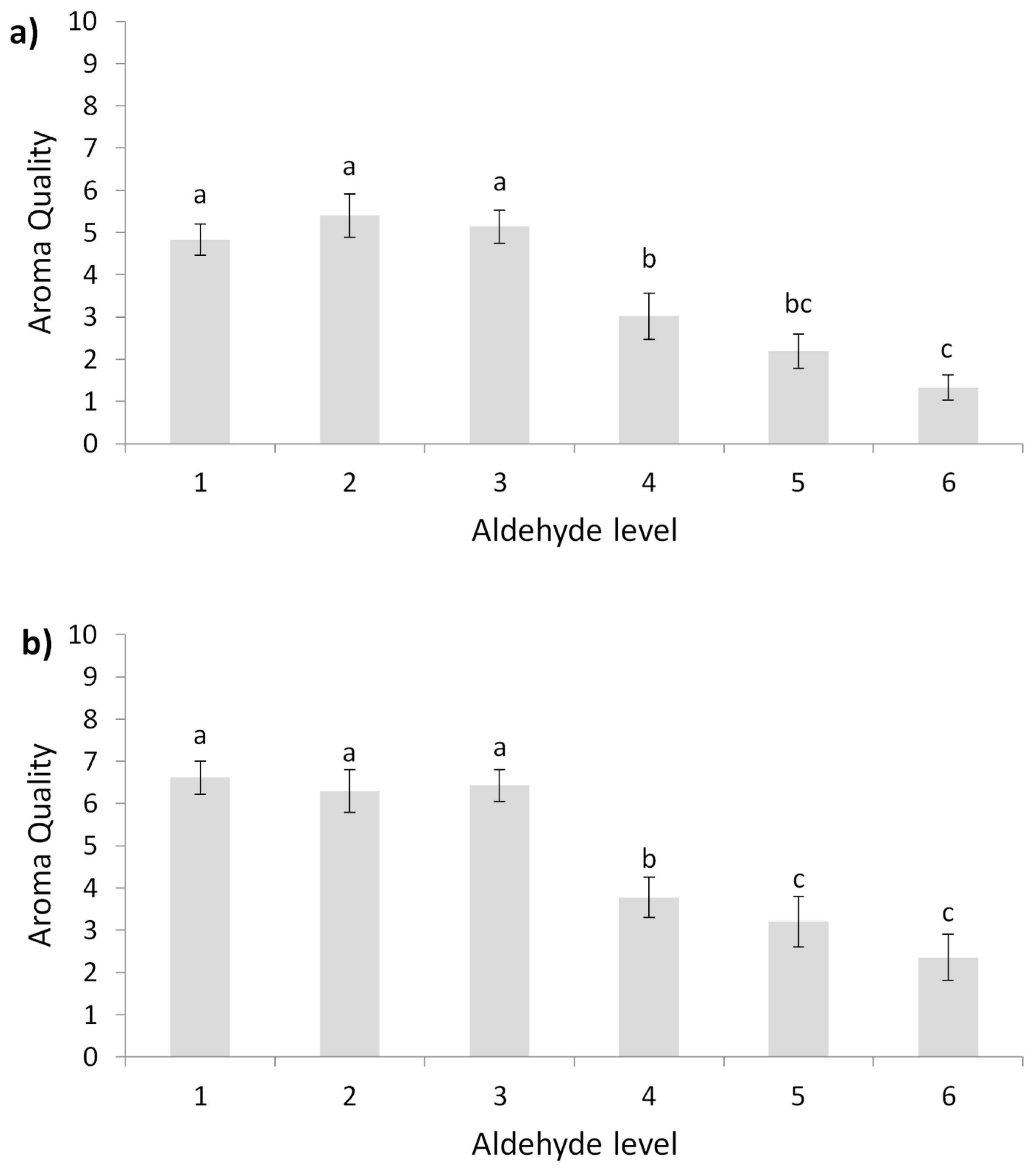
Foods | Free Full-Text | Sensory Relevance of Strecker Aldehydes in Wines. Preliminary Studies of Its Removal with Different Type of Resins
![102191-92-4 | (tert-Butyldimethylsilyloxy)acetaldehyde | Dimethyl-tert-butylsilyloxyacetaldehyde; (tert-Butyldimethylsilanyloxy) acetaldehyde; 2-[[(1,1-Dimethylethyl)dimethylsilyl]oxy]acetaldehyde; | C₈H₁₈O₂Si | TRC 102191-92-4 | (tert-Butyldimethylsilyloxy)acetaldehyde | Dimethyl-tert-butylsilyloxyacetaldehyde; (tert-Butyldimethylsilanyloxy) acetaldehyde; 2-[[(1,1-Dimethylethyl)dimethylsilyl]oxy]acetaldehyde; | C₈H₁₈O₂Si | TRC](https://www.trc-canada.com/prod-img/B942745.png)
102191-92-4 | (tert-Butyldimethylsilyloxy)acetaldehyde | Dimethyl-tert-butylsilyloxyacetaldehyde; (tert-Butyldimethylsilanyloxy) acetaldehyde; 2-[[(1,1-Dimethylethyl)dimethylsilyl]oxy]acetaldehyde; | C₈H₁₈O₂Si | TRC

PDF) DualPhos: A versatile, chemoselective reagent for two-carbon aldehyde to latent (E)-alkenal homologation and application in the total synthesis of phomolide G

A Prodrug Strategy for the In Vivo Imaging of Aldehyde Dehydrogenase Activity | Biological and Medicinal Chemistry | ChemRxiv | Cambridge Open Engage
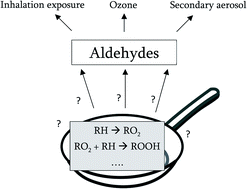
Formation pathways of aldehydes from heated cooking oils - Environmental Science: Processes & Impacts (RSC Publishing)

Formaldehyde, aliphatic aldehydes (C2‐C11), furfural, and benzaldehyde in the residential indoor air of children and adolescents during the German Environmental Survey 2014–2017 (GerES V) - Birmili - 2022 - Indoor Air - Wiley Online Library

Deoxygenative Functionalizations of Aldehydes, Ketones and Carboxylic Acids - Li - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Metal-Free Direct Deoxygenative Borylation of Aldehydes and Ketones | Journal of the American Chemical Society
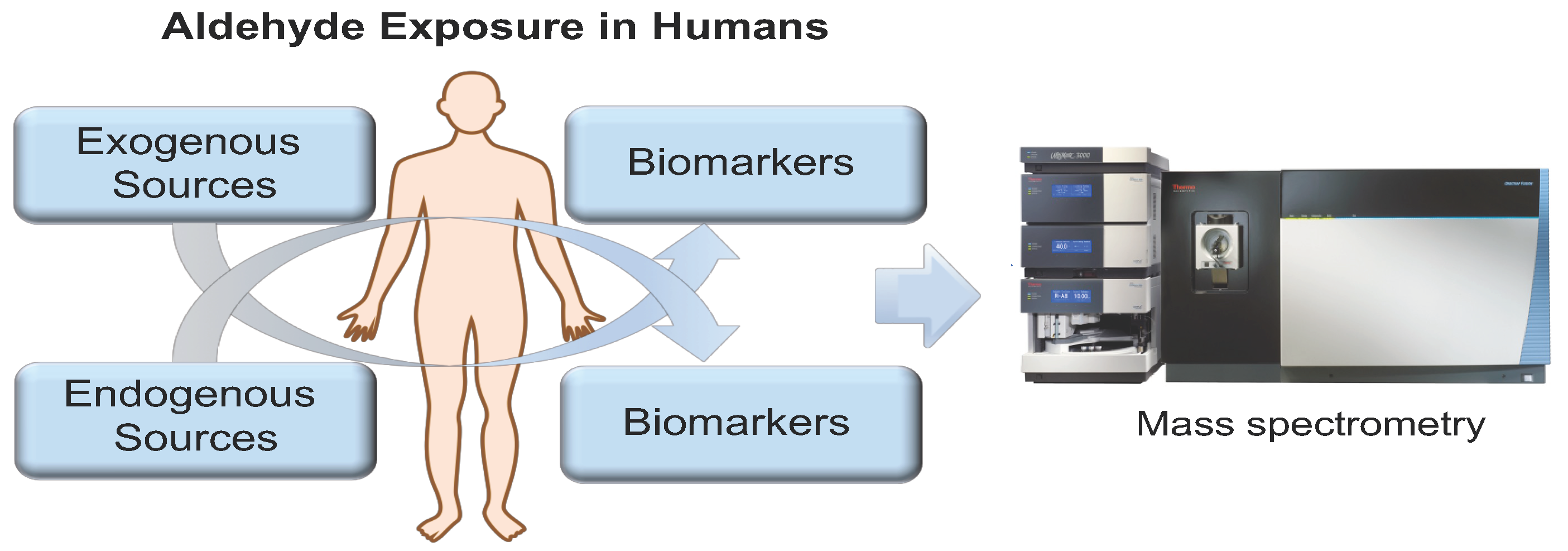
Toxics | Free Full-Text | Bioanalytical and Mass Spectrometric Methods for Aldehyde Profiling in Biological Fluids

The interaction of disulfiram and H2S metabolism in inhibition of aldehyde dehydrogenase activity and liver cancer cell growth - ScienceDirect

Here is your daily mcat study tip! Like and share this if you found it useful 🙂 #mcat #30daymcat #premed #medicine #mcatprep | Instagram

Enantioselective α-Arylation of Aldehydes via the Productive Merger of Iodonium Salts and Organocatalysis | Journal of the American Chemical Society